|

22.4 per cent of total seafood entry line refusals were of shrimp for reasons related to banned antibiotic. (Photo: Stock File)
Seventeen antibiotics-contaminated shrimp lines refused in September
 UNITED STATES
UNITED STATES
Thursday, October 12, 2017, 23:00 (GMT + 9)
Antibiotic-contaminated imported shrimp rejections from entering the United States make up nearly one-quarter of the US Food and Drug Administration’s (FDA) seafood entry line refusals in September, the Southern Shrimp Alliance (SSA) reported.
Over the first three weeks of September, the FDA reports that 17 of 76 (22.4 per cent) total seafood entry line refusals were of shrimp for reasons related to banned antibiotics.
These products had been exported by three firms, and three shrimp entwere reported by three different FDA import divisions:
- Yantai Wei-Cheng Food Co., Ltd. (China) had fourteen entry lines refused for shrimp contaminated with veterinary drug residues by the Division of West Coast Imports;
- Minh Phu Seafood Corporation (Vietnam) had two entry lines refused for shrimp contaminated with veterinary drug residues by the Division of Northeast Imports; and
- Tuta Corporation (Venezuela) had one entry line refused for shrimp contaminated with veterinary drug residues by the Division of Southeast Imports
The FDA also issued public notice of additional seafood entry line refusals in the month of August, including the identification of two more entry lines of Vietnamese shrimp refused for reasons related to banned antibiotics.
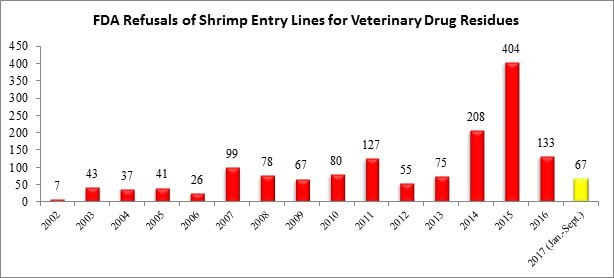
Through the first three quarters of 2017, the FDA has reported refusing 67 entry lines of shrimp for reasons related to banned antibiotics, substantially below the refusal levels reported in 2014, 2015, and 2016.
The three shrimp lines contaminated with banned antibiotics that were rejected in August came from China, Hong Kong and Vietnam.
The other two additional shrimp entry lines refused by the FDA for banned antibiotics in August were from Vietnam and had been exported by Ca Mau Seafood Processing & Service Joint Stock.
The SSA stated that the FDA’s updated reporting for the month of August omits reference to another shrimp entry line from Ca Mau Seafood Processing & Service Joint Stock, which had been reported as refused for shrimp contaminated with veterinary drug residues by the Division of Northeast Imports.
In addition, the FDA’s revised reporting additionally omits reference to a shrimp entry line for Manwill Trading Company (Hong Kong), which had been reported as refused for shrimp contaminated with veterinary drug residues by the Division of Northeast Imports.
The SSA clarified that although these two entry lines no longer appear on the FDA’s drop-down box results of seafood entry line refusals for August, they remain listed in the excel spreadsheet database of FDA entry line refusals available for download from the agency’s website.
editorial@seafood.media
www.seafood.media
|



