|

Dried Methylobacterium extorquens biomass is produced from genetically engineered Methylobacterium extorquens strain KB203
FDA authorizes single-cell protein ingredient for aquafeeds
 UNITED STATES
UNITED STATES
Tuesday, February 19, 2019, 21:30 (GMT + 9)
Aquafeed ingredient KnipBio MEal (KBM) for salmonids and other finfish species, developed by KnipBio, Inc., received the Generally Regarded As Safe (GRAS) designation from the US Food and Drug Administration (FDA) Center for Veterinary Medicine.
It is the first premium single cell protein to achieve a GRAS designation for aquafeed from the US government.
The source of protein comes from a natural leaf symbiont, Methylobacterium extorquens.As a model organism it is metabolically versatile, sharing both the nutritional features of photosynthetic microbes and the ease of use of industrially used microbes. ►
KnioBio is made with dried Methylobacterium extorquens biomass, produced from genetically engineered Methylobacterium extorquens strain KB203 through fermentation using starting raw materials suitable for use in animal food and process controls. The organism is grown in a mineral salts solution and methanol is added as the carbon source for growth. Following the fermentation, the biomass is centrifuged, and the resulting slurry is then spray dried.
“We are very excited by this development. GRAS notification status is different from self-determination because it is the result of a complex review process that takes years to complete,” said Larry Feinberg, CEO of KnipBio.
“ Many countries look to FDA GRAS designation as a strong indication that a feed product has been rigorously tested and reviewed. This designation is therefore an important milestone for our company and moves us significantly ahead of other companies in our industry," he added.
 "We are proud to be the first American company to invest in the scientific evidence necessary to receive this designation for aquafeed and are confident this approval opens many opportunities for us in the global finfish aquafeed market estimated to be worth greater than USD 10 billion annually,” he concluded. "We are proud to be the first American company to invest in the scientific evidence necessary to receive this designation for aquafeed and are confident this approval opens many opportunities for us in the global finfish aquafeed market estimated to be worth greater than USD 10 billion annually,” he concluded.
Feinberg also pointed out that the aquaculture industry will be an increasingly important provider of healthy and sustainable animal protein in the coming decades. For this to happen, the industry needs scalable and affordable alternatives to complement the finite supply of fishmeal currently used to provide protein in feed formulas.
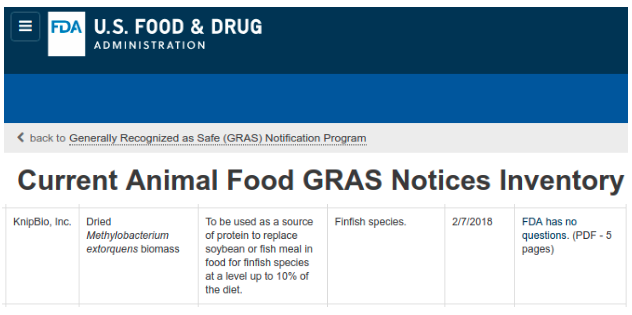
KnipBio has been incorporated into the current inventory of GRAS animal feed notices from the FDA
The company´s GRAS designation now cements KnipBio Meal as a leading protein complement to fishmeal. This milestone also provides it with opportunities to broaden industry access to its products, while eliminating any concerns about their safety and efficacy.
“For the first time, US aquafeed manufacturers will have access to a sustainable and affordable fishmeal replacement made from a traceable single cell protein," stressed Feinberg

The CEO also highlightet that after achieving GRAS designation for their first product in finfish, they now anticipate additional approvals for the use of KBM in crustaceans as well as for other versions of their single cell protein, including one that creates a natural form of astaxanthin and can potentially serve as a color additive in salmonid feed applications.
KnipBio, Inc., is based in Massachusetts. It is a pioneer in advanced nutritional solutions for animal feeds from sustainable and responsible feedstocks.
editorial@seafood.media
www.seafood.media
|



